Clinical Epidemiology Training Course
Clinical Epidemiology Training Course is designed to build advanced competencies in research methodology, clinical trials, observational studies, systematic reviews, and meta-analysis frameworks.
Course Overview
Clinical Epidemiology Training Course
Introduction
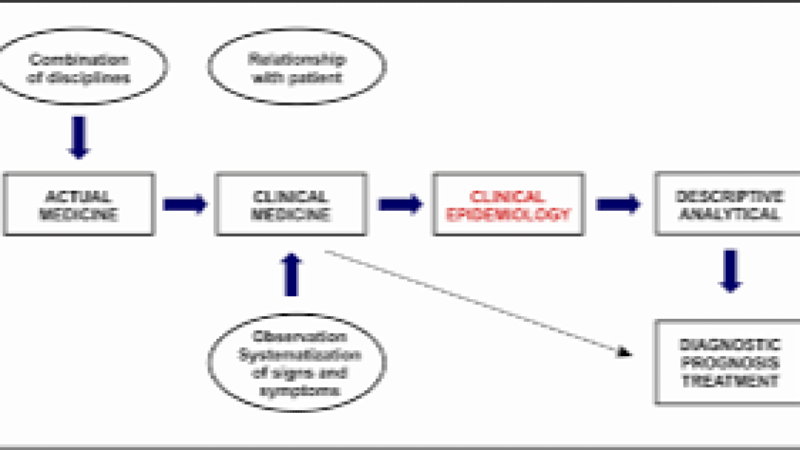
Clinical Epidemiology is the cornerstone of modern evidence-based healthcare, integrating biostatistics, population health analytics, real-world evidence (RWE), and clinical decision-making to improve patient outcomes. It focuses on applying rigorous epidemiological methods to clinical practice, enabling healthcare professionals to interpret disease patterns, risk factors, diagnostic accuracy, prognostic models, and treatment effectiveness. In the era of precision medicine, AI-driven healthcare analytics, and value-based care, clinical epidemiology has become essential for transforming raw clinical data into actionable insights.
Clinical Epidemiology Training Course is designed to build advanced competencies in research methodology, clinical trials, observational studies, systematic reviews, and meta-analysis frameworks. Participants will gain hands-on expertise in health data science, survival analysis, causal inference, and predictive modeling, empowering them to contribute to high-impact research, policy-making, and clinical innovation. The program emphasizes real-world application through case-based learning aligned with global healthcare challenges such as infectious diseases, chronic disease burden, pandemic preparedness, and health system strengthening.
Course Duration
5 days
Course Objectives
- Master fundamentals of clinical epidemiology and evidence-based medicine (EBM)
- Apply biostatistics and data interpretation in clinical research
- Understand study designs: cohort, case-control, cross-sectional, RCTs
- Develop skills in systematic review and meta-analysis (PRISMA guidelines)
- Learn causal inference and confounding bias control
- Perform risk assessment and disease modeling
- Apply survival analysis and time-to-event data techniques
- Use real-world evidence (RWE) in clinical decision-making
- Evaluate diagnostic test accuracy and ROC curve analysis
- Integrate machine learning in clinical prediction models
- Conduct outbreak investigation and infectious disease epidemiology
- Interpret healthcare data analytics and big data epidemiology
- Translate evidence into clinical guidelines and health policy
Target Audience
- Medical Doctors & Physicians
- Public Health Professionals
- Clinical Researchers & Investigators
- Epidemiologists & Biostatisticians
- Pharmacists & Clinical Pharmacologists
- Medical Students & Postgraduates
- Healthcare Data Scientists & Analysts
- Policy Makers & Health Program Managers
Course Modules
Module 1: Foundations of Clinical Epidemiology
- Principles of disease distribution & determinants
- Epidemiological measures: incidence, prevalence, risk ratios
- Introduction to evidence-based medicine
- Bias, confounding, and random error
- Case Study: COVID-19 transmission dynamics in urban populations
Module 2: Study Designs & Clinical Research Methods
- Cohort, case-control, cross-sectional studies
- Randomized controlled trials (RCTs) design
- Sampling techniques & population selection
- Internal vs external validity
- Case Study: Vaccine efficacy trial design during pandemic response
Module 3: Biostatistics for Clinical Research
- Descriptive & inferential statistics
- Hypothesis testing & p-values
- Regression models (linear & logistic)
- Survival analysis (Kaplan-Meier curves)
- Case Study: Cancer survival prediction using hospital registry data
Module 4: Systematic Review & Meta-analysis
- Literature search strategies (PubMed, Cochrane)
- PRISMA framework implementation
- Effect size calculation & heterogeneity
- Publication bias assessment
- Case Study: Meta-analysis of antihypertensive drug efficacy
Module 5: Diagnostic & Prognostic Epidemiology
- Sensitivity, specificity, predictive values
- ROC curve interpretation
- Biomarkers in clinical prediction
- Prognostic scoring systems
- Case Study: Early detection of sepsis using biomarkers
Module 6: Causal Inference & Advanced Epidemiology
- Directed acyclic graphs (DAGs)
- Confounding adjustment methods
- Instrumental variable analysis
- Counterfactual reasoning
- Case Study: Smoking and lung cancer causal pathway analysis
Module 7: Real-World Evidence & Health Data Analytics
- Electronic health records (EHR) utilization
- Big data in healthcare research
- Machine learning applications
- Predictive modeling techniques
- Case Study: AI-based diabetes risk prediction system
Module 8: Infectious Disease & Outbreak Epidemiology
- Epidemic curves & reproduction number (R0)
- Contact tracing methodologies
- Surveillance systems
- Pandemic preparedness strategies
- Case Study: Ebola outbreak containment strategy analysis
Training Methodology
This course employs a participatory and hands-on approach to ensure practical learning, including:
- Interactive lectures and presentations.
- Group discussions and brainstorming sessions.
- Hands-on exercises using real-world datasets.
- Role-playing and scenario-based simulations.
- Analysis of case studies to bridge theory and practice.
- Peer-to-peer learning and networking.
- Expert-led Q&A sessions.
- Continuous feedback and personalized guidance.
Register as a group from 3 participants for a Discount
Send us an email: info@datastatresearch.org or call +254724527104
Certification
Upon successful completion of this training, participants will be issued with a globally- recognized certificate.
Tailor-Made Course
We also offer tailor-made courses based on your needs.
Key Notes
a. The participant must be conversant with English.
b. Upon completion of training the participant will be issued with an Authorized Training Certificate
c. Course duration is flexible and the contents can be modified to fit any number of days.
d. The course fee includes facilitation training materials, 2 coffee breaks, buffet lunch and A Certificate upon successful completion of Training.
e. One-year post-training support Consultation and Coaching provided after the course.
f. Payment should be done at least a week before commence of the training, to DATASTAT CONSULTANCY LTD account, as indicated in the invoice so as to enable us prepare better for you.